- Home
- Information
- Fundamentals of radiation protection
- Physical fundamentals
- Atoms and Nuclei
Atoms and Nuclei
Bohr Model of the Atom
The Bohr model of the atom was developed in 1913 by Niels Bohr. It was the first atomic model to incorporate elements of the (then still undeveloped) quantum mechanics to gain widespread recognition.
Schematic representation of the Bohr model of the atom
In the Bohr model of the atom, atoms consist of a massive, positively charged nucleus and light, negatively charged electrons orbiting the nucleus in closed paths.
The diameter of a nucleus is about \(10^{-15}\) meters or 1 femtometer (fm).
A femtometer (fm) is a unit of length in the International System of Units equal to \(10^{-15}\) meters, or one quadrillionth of a meter. It is often referred to as "Fermi" and is the commonly used measure in nuclear physics for the diameter of atomic nuclei.
For comparison: The diameter of a human hair is about one billion (\(10^9\)) times larger than the diameter of an atomic nucleus.
The atomic nucleus is composed of positively charged protons and neutrons. Neutrons are neutral, meaning they carry no charge.
Protons and neutrons are collectively referred to as nucleons.
Nucleons are the building blocks of the atomic nucleus.
The total number of nucleons in an atom is called the mass number and largely determines the mass of the atom. Nucleons are held together by the strong nuclear force, a short-range but very strong interaction.
The diameter of the atomic shell is on the order of \(10^{-10}\) m. This size is also abbreviated as 1 Å. The size of this diameter is determined by the electrons in the outer orbits, where atoms do not have a sharp boundary but rather the electrons create a probability space.
The diameter of the atomic shell is over 10,000 times larger than the diameter of the atomic nucleus.
In describing the movement of electrons, Bohr partially set aside classical physics through three postulates. As a result, the Bohr model of the atom accurately reproduces many properties observed in the hydrogen atom, unlike older atomic models. On the other hand, many details of very precise spectroscopic measurements are still not captured by the Bohr model, and some important properties are not explained at all, including the spatial shape and the possibility of chemical bonding.
An atom with an equal number of positively charged protons and negatively charged electrons is electrically neutral.
If the number of protons in an atom is different from the number of electrons, it is called an ion. If the number of protons is greater than the number of electrons, the atom is referred to as a positively charged atom.
If the number of protons is less than the number of electrons, the atom is referred to as a negatively charged atom.
Atoms and Chemical Elements
Atoms are the basic building blocks of chemical elements.
The chemical behavior of atoms is determined by the number of protons in the nucleus and the resulting structure of the electron shell.
Examples of Chemical Elements
-
Hydrogen (H)
consists of one proton in the nucleus and one electron in the shell.
-
Helium (He)
consists of two protons in the nucleus and two electrons in the shell.
-
Oxygen (O)
consists of eight protons in the nucleus and eight electrons in the shell.
-
Uranium (U)
consists of eight protons in the nucleus and eight electrons in the shell.
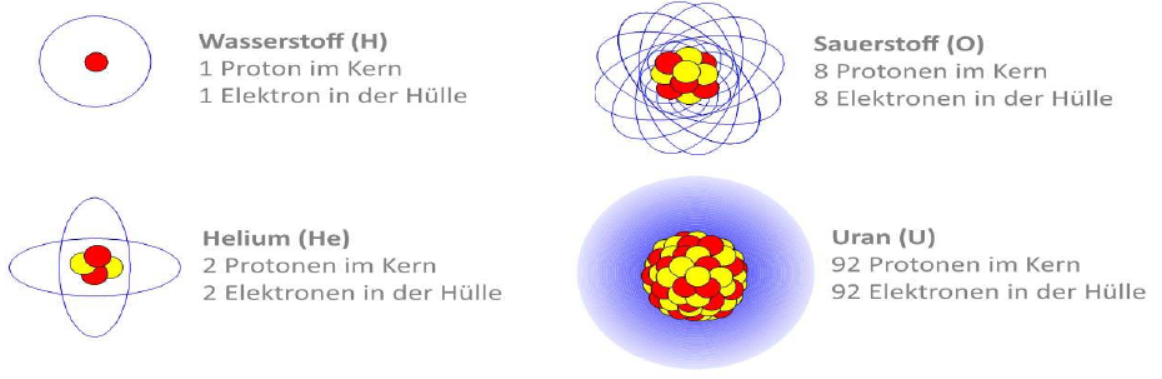
Visual representation of the atomic structure of various elements
Nuclei and Isotopes
A nucleus is any combination of protons and neutrons that forms an atomic nucleus.
A nucleus is described by the number of its protons (atomic number) and the sum of the number of its protons and neutrons (mass number).
The notation for this is \(_{A}^{M}X\). \(X\) is a placeholder for the chemical symbol (e.g., H, He, O, U).

Schematic representation of an atomic nucleus consisting of protons (red) and neutrons (blue).
More than 2000 different nuclei are known. Most of these nuclei are radioactive.
Nuclei with the same atomic number but different mass numbers are called isotopes.
Examples of Isotopes
-
Hydrogen Isotopes:
\(_{1}^{1}H\), \(_{1}^{2}H\), \(_{1}^{3}H\)
-
Carbon Isotopes:
\(_{6}^{12}C\), \(_{6}^{14}C\)
-
Uranium Isotopes:
\(_{92}^{235}U\), \(_{92}^{238}U\)
Nuclear Chart
All nuclei can be systematically represented in a nuclear chart in Segre representation.
A commonly used representation consists of a Cartesian Z,N coordinate system. The number of protons Z is plotted along the horizontal axis, and the number of neutrons N is plotted along the vertical axis.
Each known nucleus is plotted in the coordinate system according to its proton number Z and neutron number N.
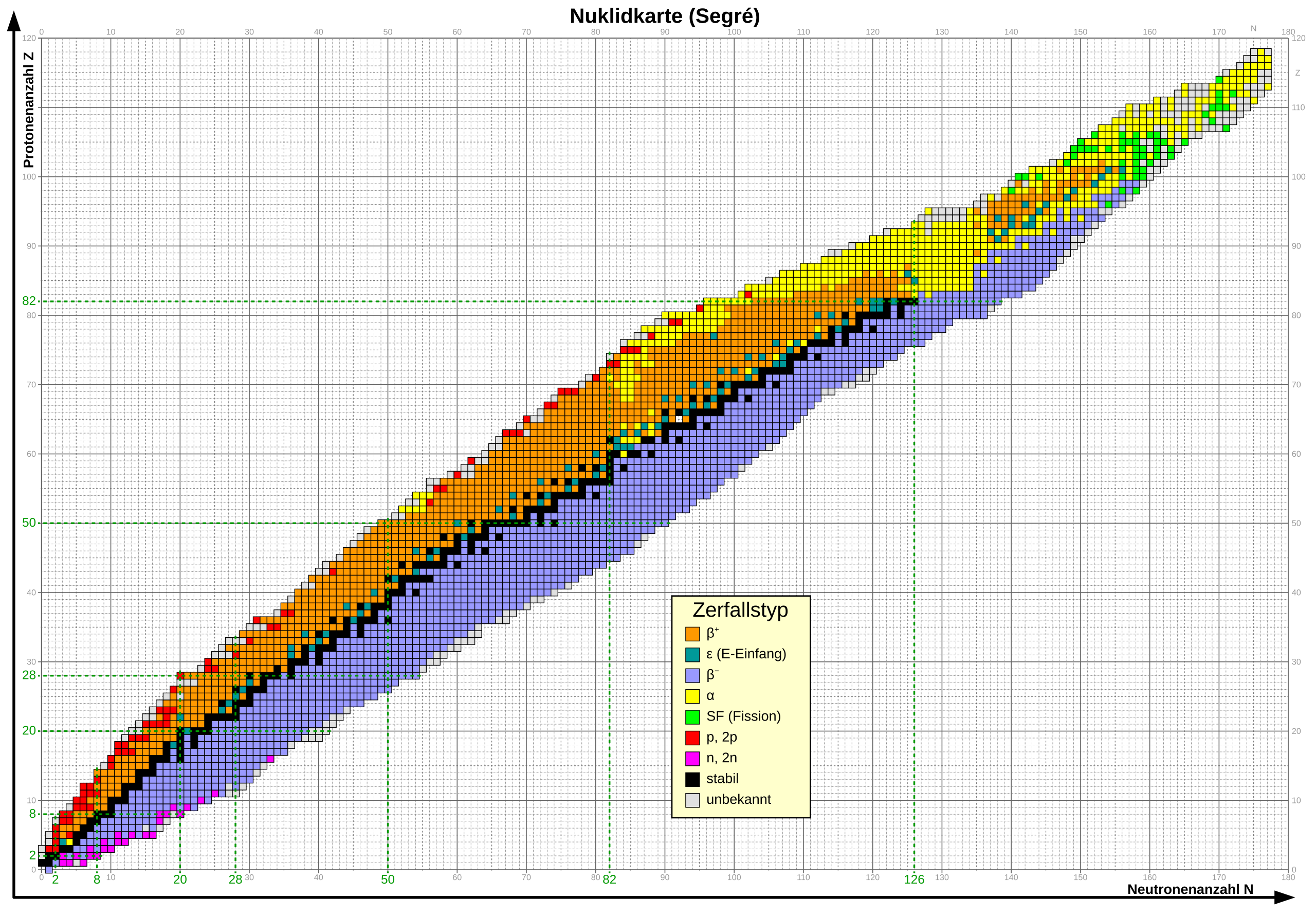
Nuclear chart in Segre representation with color coding of decay types (Nuclear chart of the Brookhaven National Laboratory).
A color coding of the respective nuclei in the nuclear chart gives indications of whether they are stable, meaning they no longer decay, or indicates their decay type. For instance, stable nuclei are often shown against a black background, while radioactive nuclei are shown against a blue (\(\beta^-\) decay), red (\(\beta^+\) decay), yellow (\(\alpha\) decay), or green (spontaneous fission) background. Additionally, the individual fields may contain information such as nucleus designation, half-life, gamma lines, etc.
The known nuclei are found along the diagonals of the Z,N plane.

Color coding used in the Karlsruhe nuclear chart for various nuclei.
Online versions of nuclear charts are available at:
