- Home
- Information
- Fundamentals of radiation protection
- Physical fundamentals
- Radiation
Radiation
What is radiation?
Radiation describes the propagation of particles and electromagnetic waves, whose common feature is the transport of energy. In the case of particles, energy is transported by so-called corpuscular radiation; in the case of electromagnetic waves, by wave radiation.
Any radiation has
- a cause, i.e. a radiation source, and
- a direction, the direction of radiation.
- radioactive substances (gamma radiation, X-ray radiation),
- mobile communications (microwave radiation), or
- lamps (light)
If radiation encounters a material—whether solid, liquid, or gaseous—i.e. matter composed of atoms or molecules, it can interact with it. These interactions can take the form of
- absorption or
- scattering
During absorption, the energy of the radiation is absorbed by the material into which the radiation penetrates. During scattering, the direction of the radiation is changed and, in addition, part of its energy may be absorbed by the material.
Transmission means the passage of radiation through matter without changing the direction of the radiation. However, some of the radiation energy may be absorbed and/or scattered on its way through matter, i.e. the energy and/or the “amount” of radiation after passing through the material may be lower than before it entered the material.
What is ionizing radiation?
Ionizing radiation is any particle or electromagnetic radiation that, through interaction with atoms or molecules, can release electrons from them or break chemical bonds, leaving positively charged ions or molecular fragments behind. This process is called ionization. Energies greater than 5 eV (electron volts) are required for this. For the chemical elements occurring in the human body, the minimum ionization energy is about 20 eV.
A distinction is made between directly ionizing and indirectly ionizing radiation. In the case of directly ionizing radiation, charged particles (e.g. electrons, protons, α particles, β particles) transfer excitation and ionization energy to atoms or molecules as they pass through matter due to their electric charge, and can thereby ionize them.
Uncharged radiation, such as electromagnetic radiation or uncharged particles, can ionize atoms or molecules by generating a charged secondary particle. In the case of gamma or X-ray radiation (these are forms of electromagnetic radiation), these may be secondary electrons; in the case of uncharged particles, such as neutrons, recoil protons.
Examples of ionizing radiation
The examples of ionizing radiation listed below all have different causes (i.e. radiation sources), but the same effect on matter.
- X-ray radiation
- Gamma radiation
- Nuclear radiation (radioactivity)
- Radiation from accelerators
- Cosmic radiation
- Neutrons
The energy unit electron volt
Energy is a physical quantity. Its practical significance often lies in the fact that a physical system can release heat, perform work, or emit radiation to the extent that its energy decreases. Energy is expressed in joules (J).
In connection with ionizing radiation, energy is stated using a special unit, the electron volt (eV).
The relationship between the units joule and electron volt is given by
1 eV = 1.6·1019 J
This means that an electron volt represents very small energies compared to a joule.
For further understanding:
A joule is the energy needed, for example, to lift a body with a mass of about 102 grams by one meter (note: this requires a force of one newton).
The quantity 1 eV can also be described intuitively: it is the energy of motion (kinetic energy) that an electron acquires when it passes through a potential difference of U = 1 eV in a vacuum.
Schematic illustration of the meaning of one electron volt (eV) as the energy an electron acquires when it passes through a potential difference of one volt (V) in a vacuum.
As we will see in a moment, the different types of radiation cover a very wide energy range. Since one wants to avoid numbers with too many digits when specifying energy values, the use of so-called prefixes for units has proven extremely practical. Here, a letter (the so-called prefix) is placed in front of the unit eV. In everyday life, we already use these prefixes almost automatically, for example when stating masses. Instead of 1000 g, we usually say 1 kg. Here, the prefix k is placed before the symbol for the unit gram (g). k means that the stated value in the base unit (here g) is one thousand times greater than the numerical value.
Because the various types of radiation can cover a very large energy range and one wants to avoid numbers with too many digits when specifying energy values, the use of so-called prefixes for units has proven extremely practical. Here, a letter (the so-called prefix) is placed in front of the unit eV. This is familiar from practical experience, for example when stating masses. 1000 g can be written more briefly as 1 kg.
Some of the prefixes most commonly used in connection with radiation are listed below:
| Prefix | Name | Value (number) | Value (power of ten) | Value (word) |
|---|---|---|---|---|
|
µ
|
Micro
|
0,000001
|
\(10^{-6}\)
|
Millionth
|
|
m
|
Milli
|
0,001
|
\(10^{-3}\)
|
Thousandth
|
|
k
|
Kilo
|
1000
|
\(10^{3}\)
|
Thousand
|
|
M
|
Mega
|
1000000
|
\(10^{6}\)
|
Million
|
|
G
|
Giga
|
1000000000
|
\(10^{9}\)
|
Billion
|
|
T
|
Tera
|
1000000000000
|
\(10^{12}\)
|
Trillion
|
|
P
|
Peta
|
1000000000000000
|
\(10^{15}\)
|
Quadrillion
|
|
E
|
Exa
|
1000000000000000000
|
\(10^{18}\)
|
Quintillion
|
Examples of energy ranges for different types of radiation
With knowledge of the meaning of the prefixes, the typical energy ranges for the different types of radiation listed below can be understood more easily.
| Type of radiation | Energy range |
|---|---|
|
X-ray radiation
|
~10 keV to 300 keV
|
|
Gamma radiation
|
~100 keV to 3 MeV
|
|
Alpha radiation
|
~3 MeV to 10 MeV
|
|
Medical accelerators
|
up to 25 meV
|
|
Cosmic radiation
|
up to \(10^{14}~MeV = 100~TeV\)
|
Ionizing radiation and other types of radiation
A graphical representation of the various regions of the electromagnetic spectrum is shown in the figure below. The typical classifications of the different radiation regions and their corresponding designations are shown there according to wavelength and frequency. The red values are the corresponding energy values.
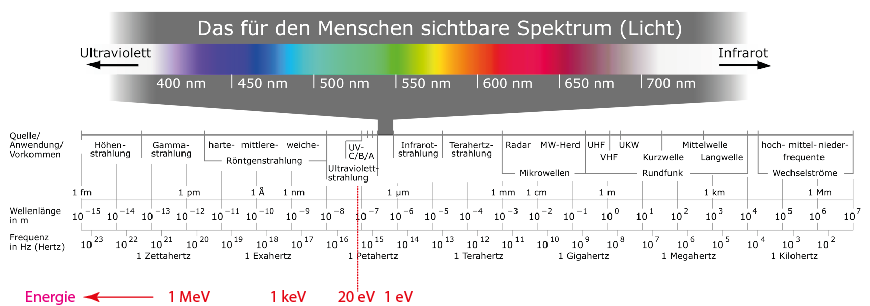
Overview of the different regions of the electromagnetic spectrum (from https://de.wikipedia.org/wiki/Elektromagnetisches_Spektrum). In addition, various energy values (in red) have been added.
Note:
The “size” of electromagnetic radiation can be expressed as energy (unit eV), wavelength (unit m), or frequency (unit Hz), since these quantities are directly related to one another by formulas, i.e. a value given in eV can be converted into the corresponding value in m or Hz. A suitable online calculator can be found, for example, at https://rechneronline.de/spektrum/.
Here is a short overview of the corresponding energy, wavelength, and frequency values determined with the online calculator:
| Energy | Frequency | Wavelength |
|---|---|---|
|
1 eV
|
241.8 THz
|
1.2 µm
|
|
20 eV
|
4.8 PHz
|
62 nm
|
|
1 keV
|
241.8 PHz
|
1.2 nm
|
|
1 MeV
|
241.8 EHz
|
1.2 pm
|
Important types of ionizing radiation
In practical radiation protection, we essentially have to consider four types of radiation.
- the electrically charged alpha and beta particles, as well as
- the electrically neutral photons and neutrons.
Their most important properties are briefly summarized below.
Alpha particles (\(\alpha\) particles)
It consists of 2 protons and 2 neutrons and has no electrons. It therefore carries a double positive charge.
Beta particles (\(\beta\) particles)
Beta particles can occur in two forms. On the one hand as electrons, which carry a negative elementary charge, and on the other hand as positrons, which carry a positive elementary charge.
Electrons and positrons are almost identical. They differ only in their charge, which is negative for electrons and positive for positrons. The positron is also referred to as the antiparticle of the electron. When an electron and a positron meet, they annihilate each other (so-called annihilation) and convert their mass completely into radiation.
Photons
Photons can occur in the form of X-ray or gamma radiation. X-ray radiation originates in the electron shell of a nuclide, gamma radiation in the atomic nucleus.
Photons are electromagnetic radiation that has no charge and no (rest) mass.
Neutrons
Neutrons are components of the nucleus and can be released, for example, during nuclear fission. A neutron is about 1839 times heavier than an electron and has no electric charge.
Ranges of ionizing radiation
From the perspective of active radiation protection, it is important to know the ranges of the different types of radiation in air. This already allows an initial assessment of whether radiation protection measures are required at all and, if so, which ones. For example, if the range of radiation from a source were only a few millimeters, but the smallest distance that a person could assume from that source under any conceivable circumstances were in the range of meters, radiation protection measures could be dispensed with.
Let us now consider the four types of radiation discussed above with regard to their typical ranges. It should be noted that these depend on the energy of the radiation. The higher the energy, the farther the radiation reaches.
| Radiation | Range in air | Range in tissue |
|---|---|---|
|
Alpha radiation
|
a few centimeters |
a few micrometers |
|
Beta radiation |
at most a few meters
|
a few millimeters
|
|
Photons |
many meters
|
many meters
|
|
Neutrons |
almost unlimited
(many meters) |
in some cases very long range
(depending on neutron energy) |
Radiation energy and radiation intensity
Two other relevant quantities in practical radiation protection are the concepts of radiation energy and radiation intensity.
The term radiation energy refers to the energy possessed by the radiation, i.e. a particle, a photon, or a neutron. Among other things, the type and strength of the interaction of radiation with matter are determined by the magnitude of this energy.
Radiation energies that occur, for example, during radioactive decay are the characteristic energies of the emitted gamma radiation or particles. Some examples are listed below:
Gamma radiation
| Isotope | Name | Energy |
|---|---|---|
|
\(^{241}Am\)
|
Americium-241
|
\(59.5~keV\)
|
|
\(^{137}Cs\)
|
Cesium-137
|
\(661.7~keV\)
|
|
\(^{60}Co\)
|
Cobalt-60
|
\(1173.2~keV\)
|
|
\(^{60}Co\)
|
Cobalt-60
|
\(1332.5~keV\)
|
Note: radioactive isotopes can emit gamma radiation with different characteristic radiation energies (see \(^{60}Co\)).
Alpha radiation
| Isotope | Name | Energy |
|---|---|---|
|
\(^{226}Ra\)
|
Radium-226
|
\(4.78~MeV\)
|
|
\(^{232}Th\)
|
Thorium-232
|
\(4.01~MeV\)
|
|
\(^{228}Th\)
|
Thorium-228
|
\(5.42~MeV\)
|
|
\(^{235}U\)
|
Uranium-235
|
\(4.392~MeV\)
|
But it is not only radiation energy that plays a role, but also its “amount”. This is defined by the term radiation intensity.
Radiation intensity denotes the number of particles, photons, or neutrons that pass through a certain area within a certain time interval. For better comparability of different values, the area is related to a unit area, often 1 cm2 or 1 m2. The time interval is often related to 1 s. This results in the unit of radiation intensity cm-2·s-1 or m-2·s-1. This unit also gives rise to the alternative term particle flux density.
If the radiation intensity also takes into account the energy transported per unit area and time, this is referred to as energy flux density. The unit is then eV·cm-2·s-1 or eV·m-2·s-1, i.e. energy per area and per unit time.


