- Home
- Measurement methods
- Gamma-Spectrometry
- Beginner
- Gamma Spectrum
Gamma Spectrum
Let’s start with a brief summary of what we’ve learned so far in this section.
During radioactive decay, isotopes decay into other stable or also radioactive isotopes. During this decay, gamma radiation can be emitted. The energies of this gamma radiation are characteristic of the decaying isotope. With a suitable detector, the energy of the gamma radiation can be determined. Its distribution, i.e., how often gamma radiation of a certain energy has been measured, can be displayed in a spectrum. In individual channels, we will then see increases (lines), called peaks, that correspond to the characteristic energies of the respective isotopes.
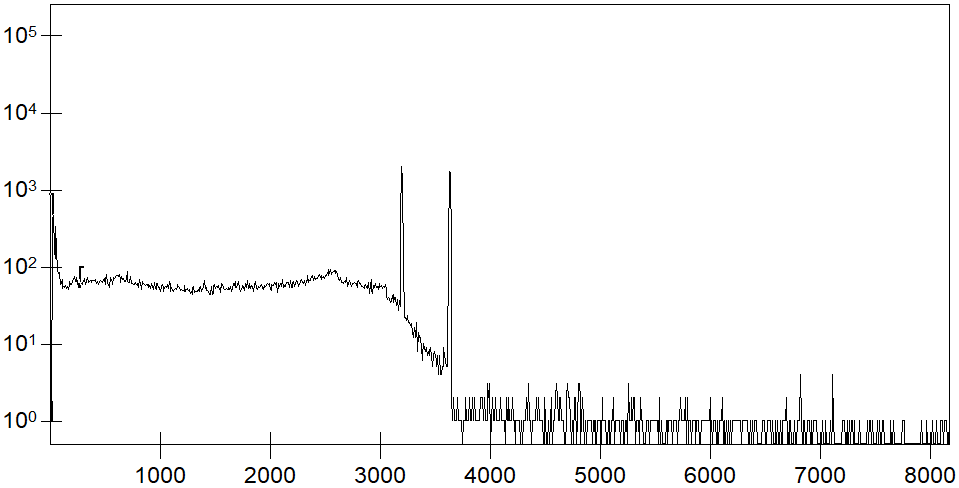
Example of a gamma spectrum. Displayed is the frequency with which the energy of a corresponding channel number of the gamma radiation was registered in the detector.
Did you notice anything in the summary that we haven’t described precisely?
Take another look at the last sentence: it states that in certain channels, the lines appear that correspond to the characteristic energies of the respective isotope. That’s correct! However, our actual task in gamma spectrometry is that we usually do not know the isotope and want to determine it through gamma measurement.
The “problem” here is that we can choose the number of channels in the multichannel analyzer and the various settings in the detector electronics (Black-Box) almost freely. This means that depending on the chosen settings, the same characteristic lines will appear in different channels in the spectrum. Thus, we generally cannot say that, for example, the line at channel number 352 corresponds to an energy of 121 keV. We still lack a link between channel number and energy.
And that leads us to the calibration of a detector system, the energy calibration.
Energy Calibration
Energy calibration addresses the mentioned problem of assigning channels in a spectrum to energies. However, it should be noted that for any changes to the settings of the detector system, i.e., changing the number of channels, the settings of the detector electronics, etc., a new energy calibration is required.
In practice, most measurement sites are used for many weeks or months with unchanged settings. Therefore, a regular check of the energy calibration is sufficient.
But how does an energy calibration actually work?
If you have read the earlier explanations thoroughly, you should already know (or at least have an inkling). When you conduct measurements with known isotopes (e.g., commercially available calibration sources), you can extract the energies of the gamma radiations of the isotopes from the corresponding data sheets or databases.
It is easiest to evaluate the measurements if you use isotopes that emit only one or two characteristic lines, such as 241Am (at 56 keV), 137Cs (at 661 keV), or 60Co (at 1173 keV and 1332 keV). You then perform a measurement (i.e., recording the distribution) with one of the isotopes and note the channel numbers where you “see” its lines.
We have already shown examples of what such spectra can look like in various ways. For a measurement with the nuclide 60Co, we will show the spectrum again below.
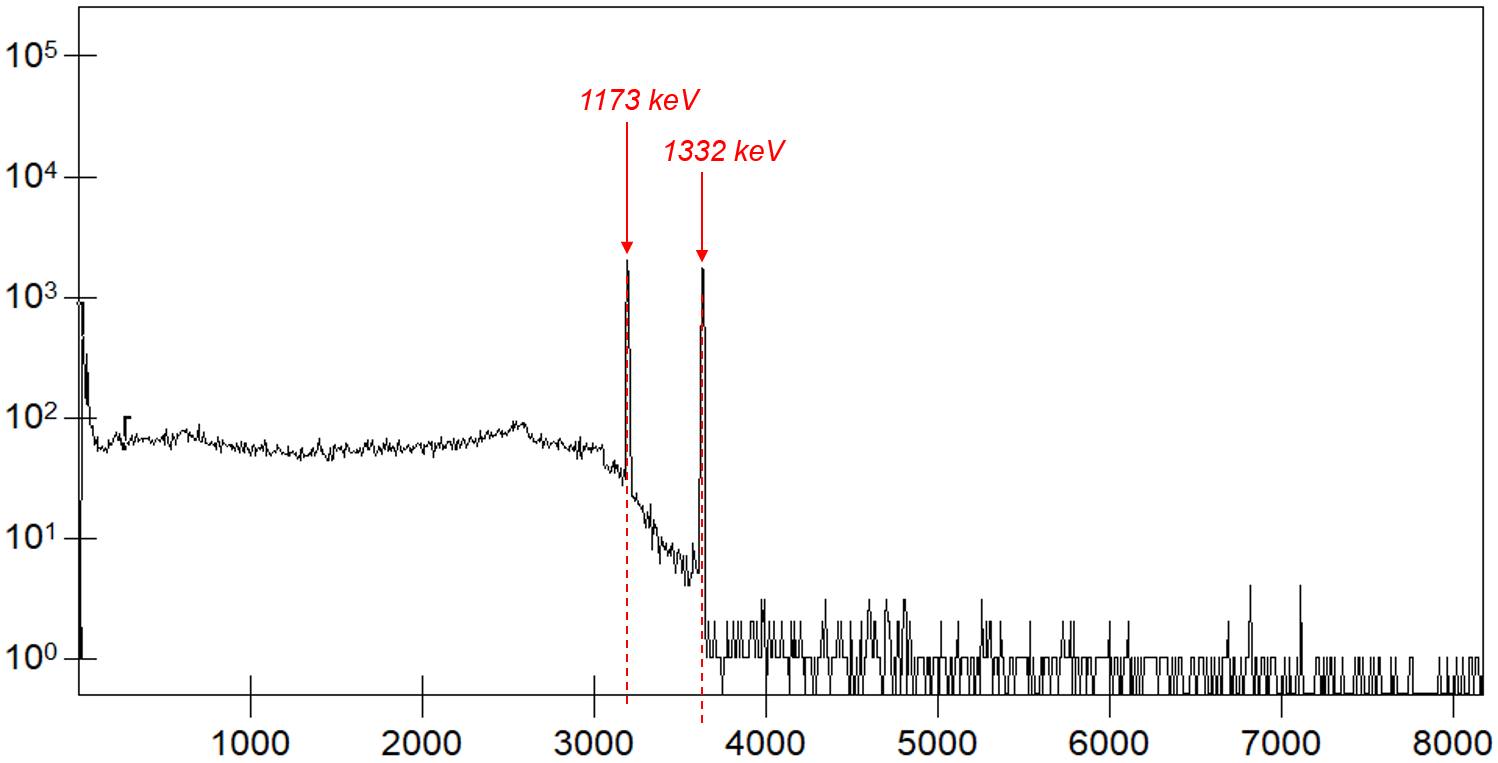
Spectrum measured with a 60Co calibration source. The two corresponding peaks at 1173 keV and 1332 keV are marked, and the corresponding channel numbers can be read on the horizontal axis (dashed lines).
Repeat this with the other isotopes and enter the respective values (energy and channel number) in a table.
The result of these measurements might look as follows (Note: the values indicated are not from the spectra shown above!):
| Channel Number | Energy in keV |
|---|---|
|
286 |
59.5 |
|
3582 |
661.7 |
|
6382 |
1173.3 |
|
7253 |
1332.5 |
The noted assignments of channel number – energy can now be plotted in a graph, as shown below. In the graph, the horizontal axis represents the channel number, and the vertical axis represents the energy in keV (the units of energy – here keV – and their significance will be discussed in a later section).
The dashed lines illustrate, for example, how the point for the gamma energy at 661.7 keV and the corresponding channel number 3582 is determined: first, a horizontal line (green dashed) is drawn, which intersects the energy axis (vertical axis) at 661.7 keV, then a vertical line (blue dashed) is drawn, which intersects the channel number axis (horizontal axis) at the value 3582. The intersection point of the two dashed axes is the sought point (marked in red). Similarly, further points can be generated.
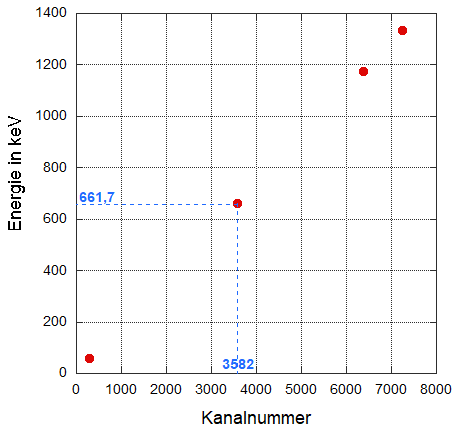
Diagram with plotted pairs of values (channel number, energy) from the energy calibration measurements. The isotopes used were 241Am, 137Cs, and 60Co. The dashed line exemplifies how the calibration point for the characteristic line of 137Cs is determined.
Now, draw a straight line through the individual points, which will allow you to determine the corresponding energy for each channel number.
If you now “see” a characteristic line in a measured spectrum, determine its channel number and use the graph to find the corresponding energy. In the following diagram, this is shown for channel number 4500, which is assigned an energy of 829.4 keV.
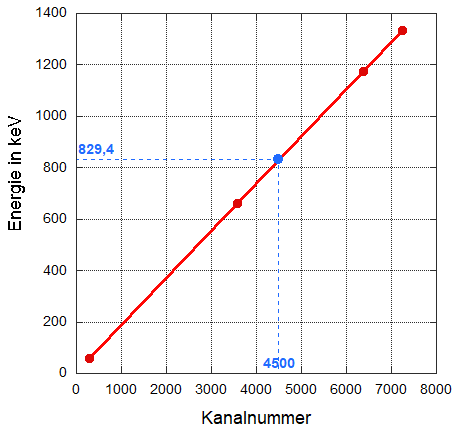
And now the diagram with the plotted (calibration) line that runs through the pairs of values. The dashed lines clarify how the corresponding energy can be determined for a channel number.
Note:
This procedure only provides correct values if the settings of the detector system during energy calibration are identical to the settings of the subsequent measurements!
In practice, such a diagram is “only” used for visualizing an energy calibration. The straight line (the so-called calibration line) is then described by a mathematical equation, whose parameters are automatically determined by fitting the values of the energy calibration. To determine an energy, only the channel number needs to be plugged into the formula. This is, of course, much faster, more precise, and thus more effective.
Since the relationship between channel number and energy is not always linear (i.e., a straight line), deviations in the relationships can also be described through mathematical formulations.
In gamma spectrometry, suitable gamma spectrometry programs are used for measurement and evaluation. These can take into account the results of existing energy calibrations in the representations of the measured spectra, i.e., the values on the horizontal axis, which originally contained the channel numbers of the multichannel analyzer, are automatically converted into the corresponding energy values. This way, the energy of a line can be easily extracted from the spectrum.
The following illustrations exemplify the representation of gamma spectra in various (commercial) programs.
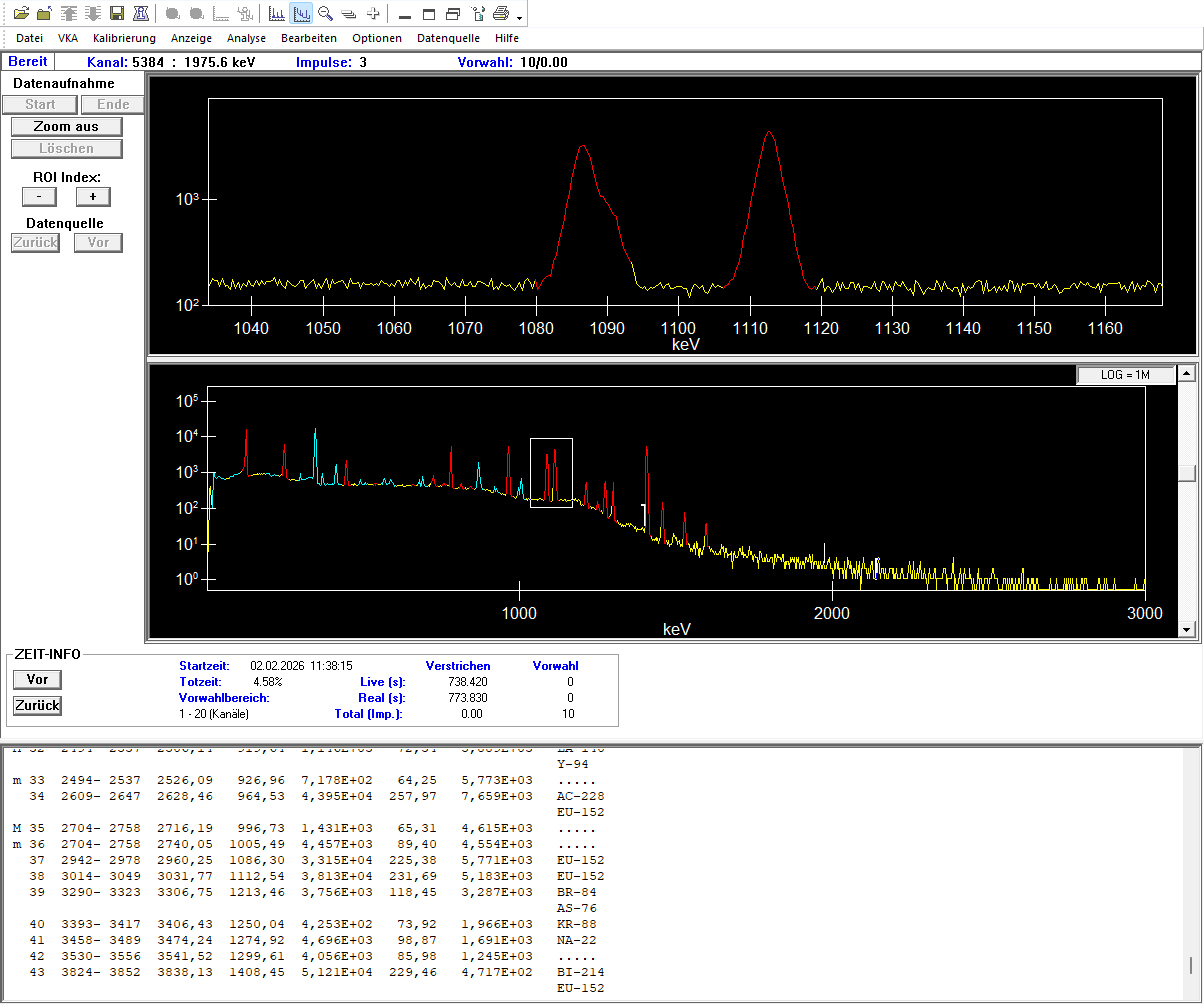
Gamma spectrum of a 152Eu sample shown in the commercial gamma spectroscopy program Genie 2000 by Mirion. The upper spectrum is an enlarged section of the entire spectrum shown below (white box). The identified peaks are marked in red in both spectra, with their data listed in the report beneath the spectra.
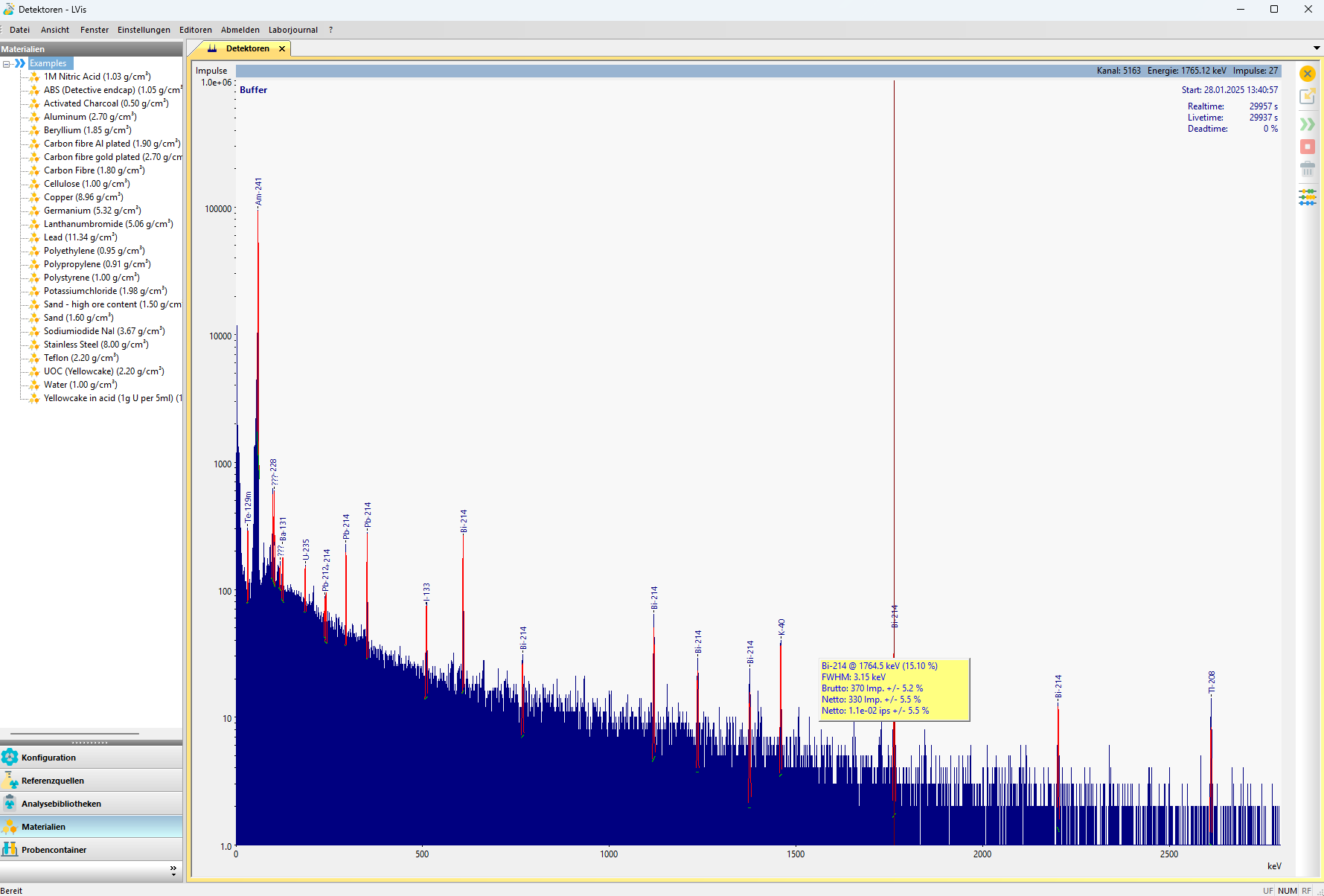
Gamma spectrum of a uranium sample shown in the commercial gamma spectrometry program LVis by Ortec. The identified peaks from uranium and radionuclides of the associated decay series are marked in red. The left window lists defined materials for use in the evaluation.
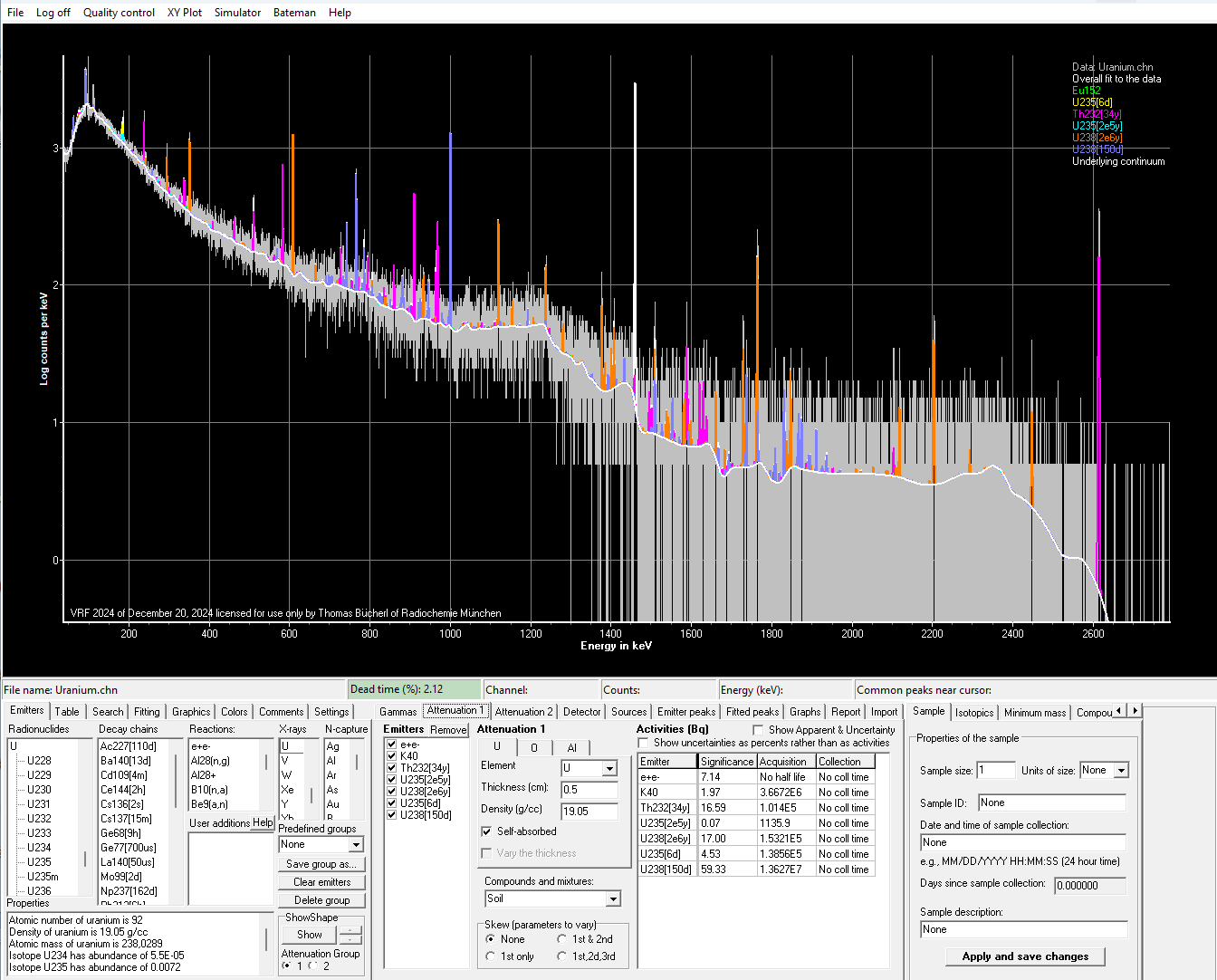
Gamma spectrum of a uranium sample shown in the commercial gamma spectrometry program VRF by Snakedance. The evaluation, i.e., identification of all peaks, is not yet complete. The radionuclides identified so far are shown in color in the spectrum, and their assignment is displayed in the legend at the top right. The white solid line shows the determined background.
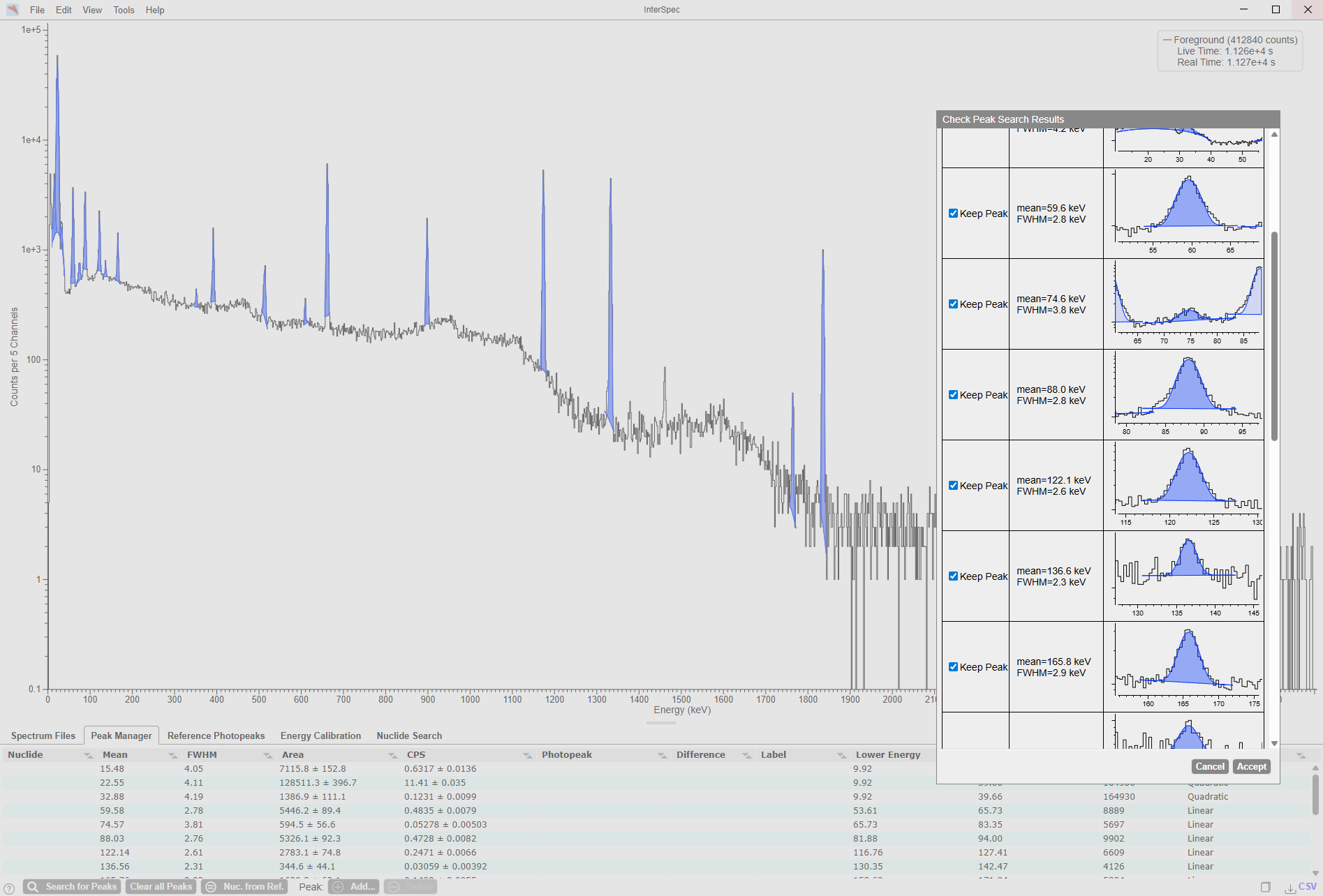
Gamma spectrum of a QCY sample, a mixture of various radionuclides for calibration measurements, shown using the gamma spectroscopy program available under the LGPL-2.1 License InterSpec. The spectrum displays automatically identified radionuclides along with a selection window for adopting the results. In the lower area, the peak manager is shown.
Now you know how to establish the link between channel number and corresponding energy through energy calibration. We are now ready to begin with the identification of radionuclides based on their characteristic lines in the measured spectrum.


