- Home
- Measurement methods
- Gamma-Spectrometry
- Beginner
- Nuclide Identification
Nuclide Identification
As already mentioned and also evident from the various representations of spectra, every radioactive isotope that emits gamma radiation shows a distinct set of energy peaks (usually just called peaks) in its spectrum. If the measurement conducted has a correct energy calibration, we only need to identify all the peaks in the spectrum and note their corresponding energies. Once we are done, we take the list of energy values and check in a database or a catalog which isotope corresponds to each energy, and we have completed the nuclide identification.
OK, so much for the theoretical approach! In practice, it is often somewhat more complicated – or we simply rely on the automatic peak identification of the gamma spectrometry software used. However, these do not always work flawlessly, and we should always at least check their results for plausibility.
Note:
To avoid misinterpretations of the statement that the programs do not always work flawlessly, it should be explicitly mentioned here that we do not mean program errors (bugs), but influences that lie outside the program and can affect the results of the (automatic) evaluation. These can be caused, for example, by poor statistics, overlaps of peaks, etc. But more about this later in the Advanced section.
The human observer usually finds it very easy to recognize peaks in a spectrum, but automation using computers is not trivial. Therefore, there exists a large number of different approaches or algorithms that serve this purpose.
Here, we first want to avoid relying on automatic evaluations and manually tackle the peak identification and describe the basic procedure. Subsequently, we will have the tools to verify the results of the automatic evaluation. However, we first need to ask ourselves the questions
- What is a peak, anyway?
- What shape can a peak have?
You probably already know, but here is the explanation, for completeness, from Wikipedia:
The term peak comes from English and means "summit, tip, vertex". In measurement technology and stochastic, a peak denotes a significant peak value.
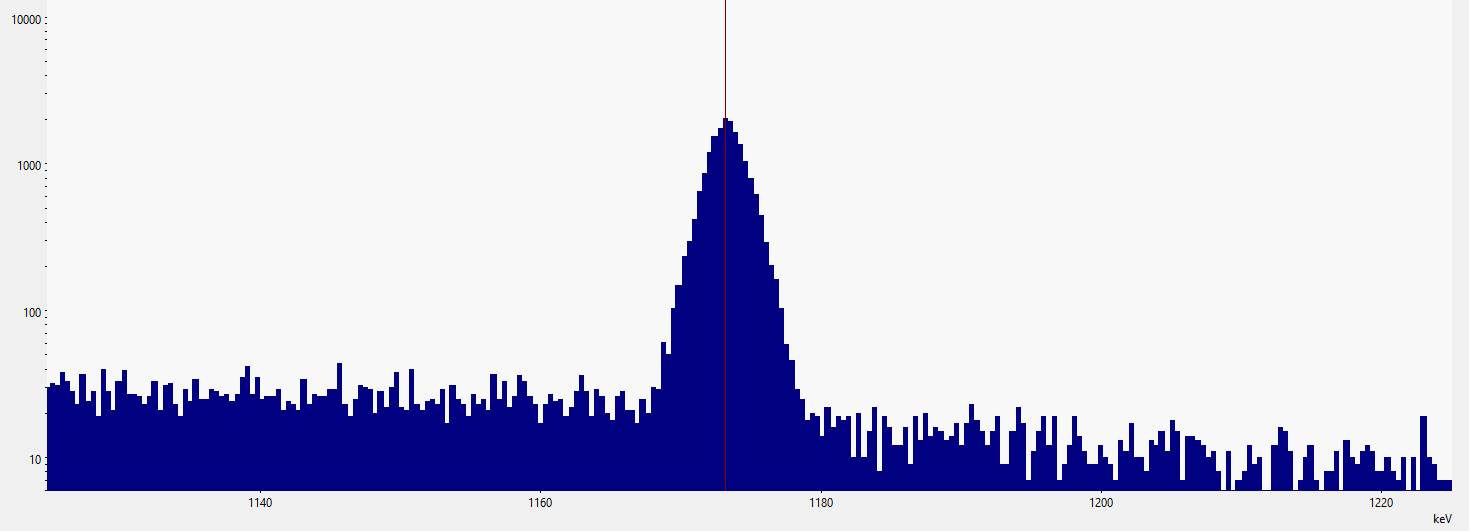
Example of what a typical gamma peak looks like in a gamma spectrum measured with an HPGe detector (High Purity Germanium Detector). The "width" of the individual channels is responsible for the stepwise representation of the peak.
In this context, the indication "significant" is important. And this is precisely where the great challenge lies for all automatic peak determinations: when is a peak significant?
Let’s consider the following three illustrations. In all three cases, the measurement setup is identical and the same nuclide (137Cs) was measured, but for different measurement times.
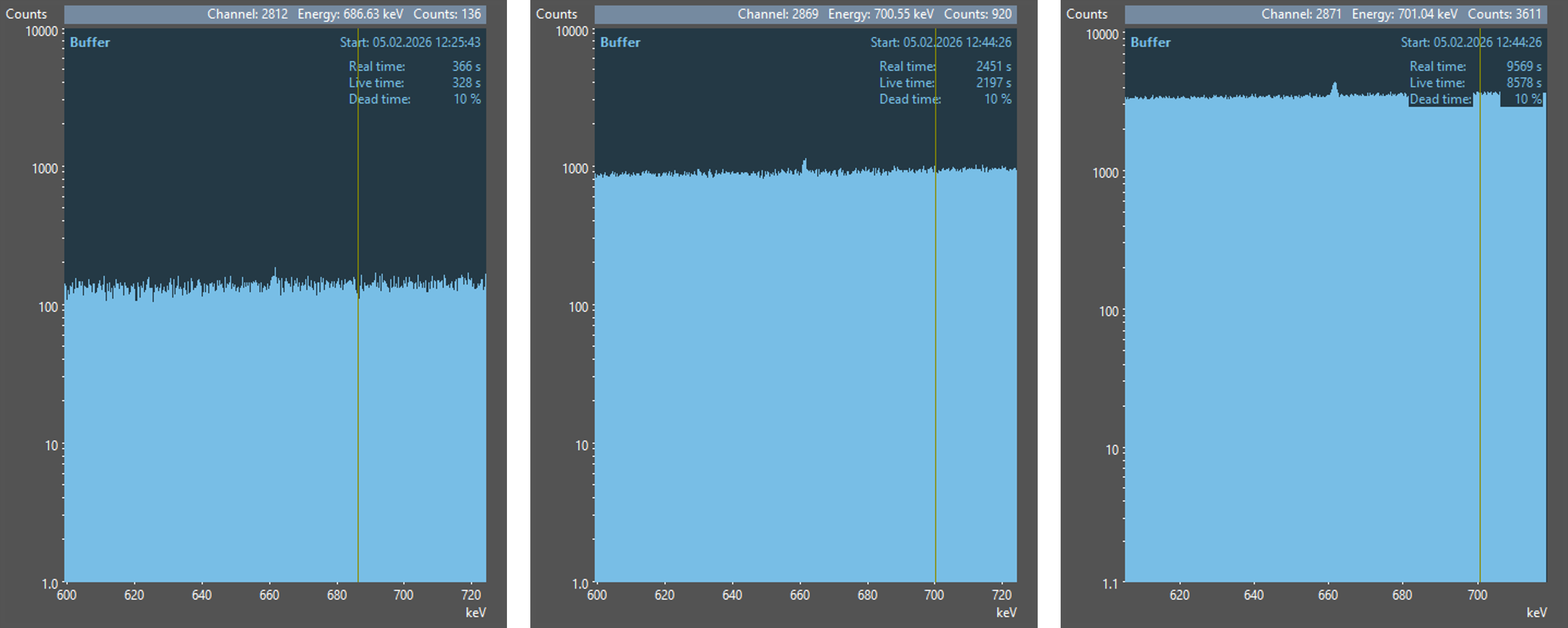
Examine the three illustrations and try to find peaks! And now look at the results of an automatic peak search with gamma spectrometry software.
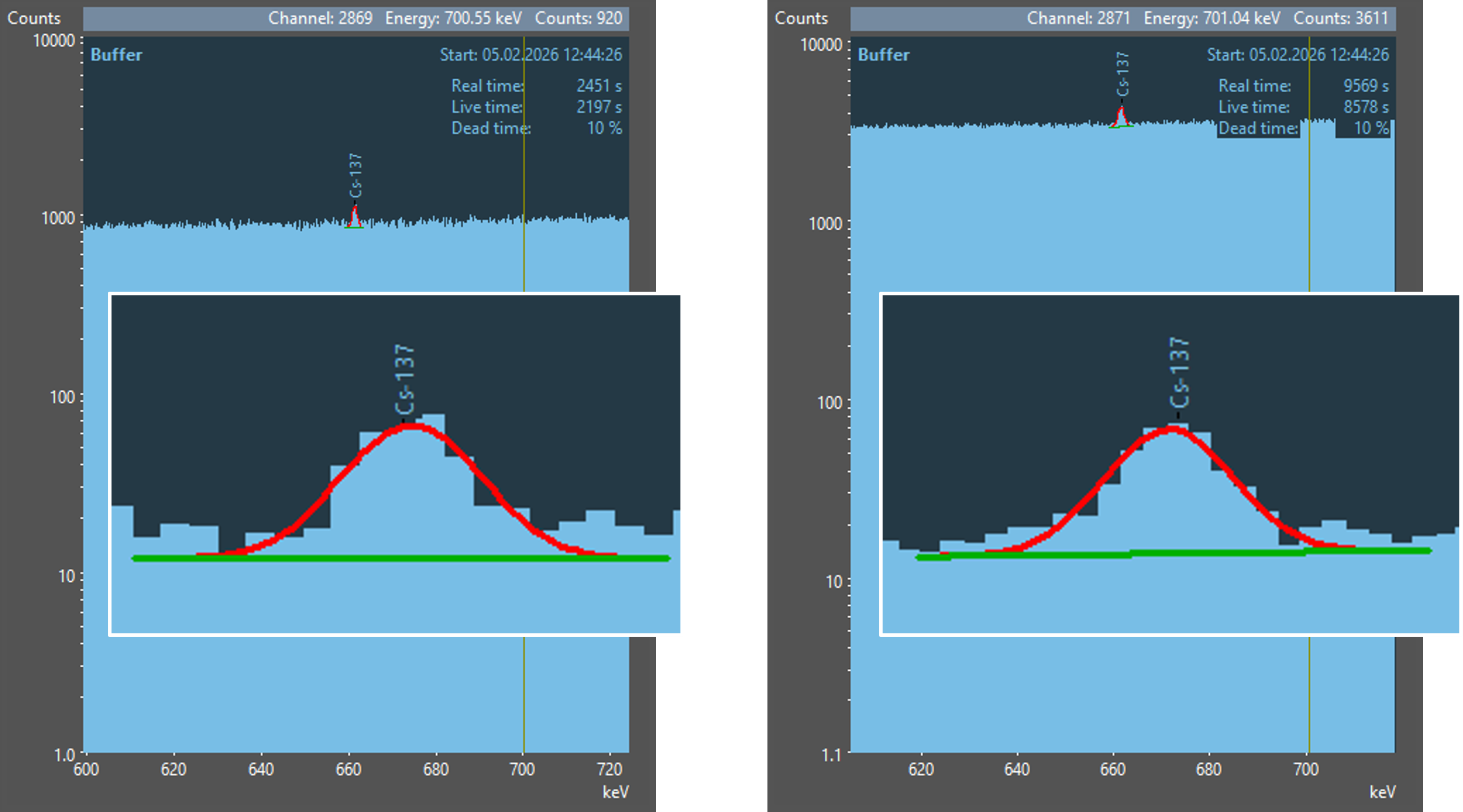
In the first spectrum, no peak was found by the automatic peak search. Did you spot one there? If so, then you should also be aware of the enormous capabilities of the human brain! In the other two spectra, the peak from 137Cs was found and fitted with a corresponding curve (red line). Additionally, an enlarged view of the peak was overlaid on the spectra for better visibility.
Information:
Currently, numerous development projects in the field of artificial intelligence (AI) and specifically machine learning (ML) are also focusing on automatic peak detection in gamma spectra.
Now, let's turn to the second question, the peak shape. But why is this question even relevant once we have found a peak?
The question is easily answered: On the one hand, we are anticipating a later step in the evaluation of gamma spectra, which is quantification (see section Advanced), and on the other hand, we need this information for identification. In both cases, we need the area contents under the peaks.
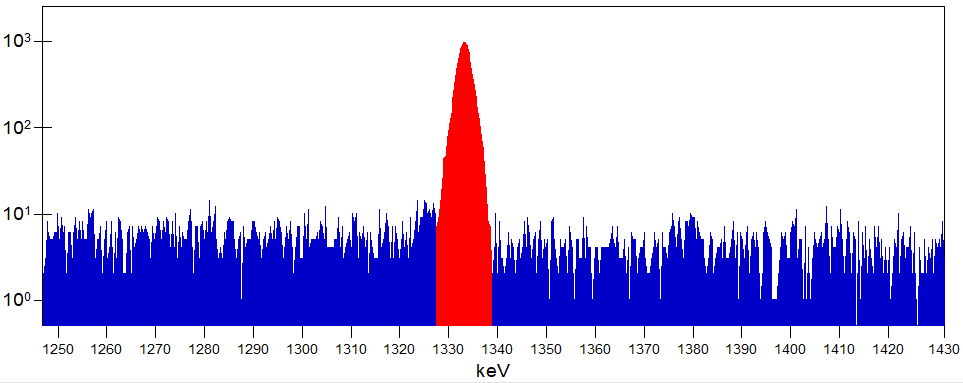
Excerpt of a gamma spectrum with highlighted area of a peak.
Area determination is usually carried out by a fit of a function that describes the peak shape and thus the physical effects that led to its formation as accurately as possible, to the data of the measured spectrum.
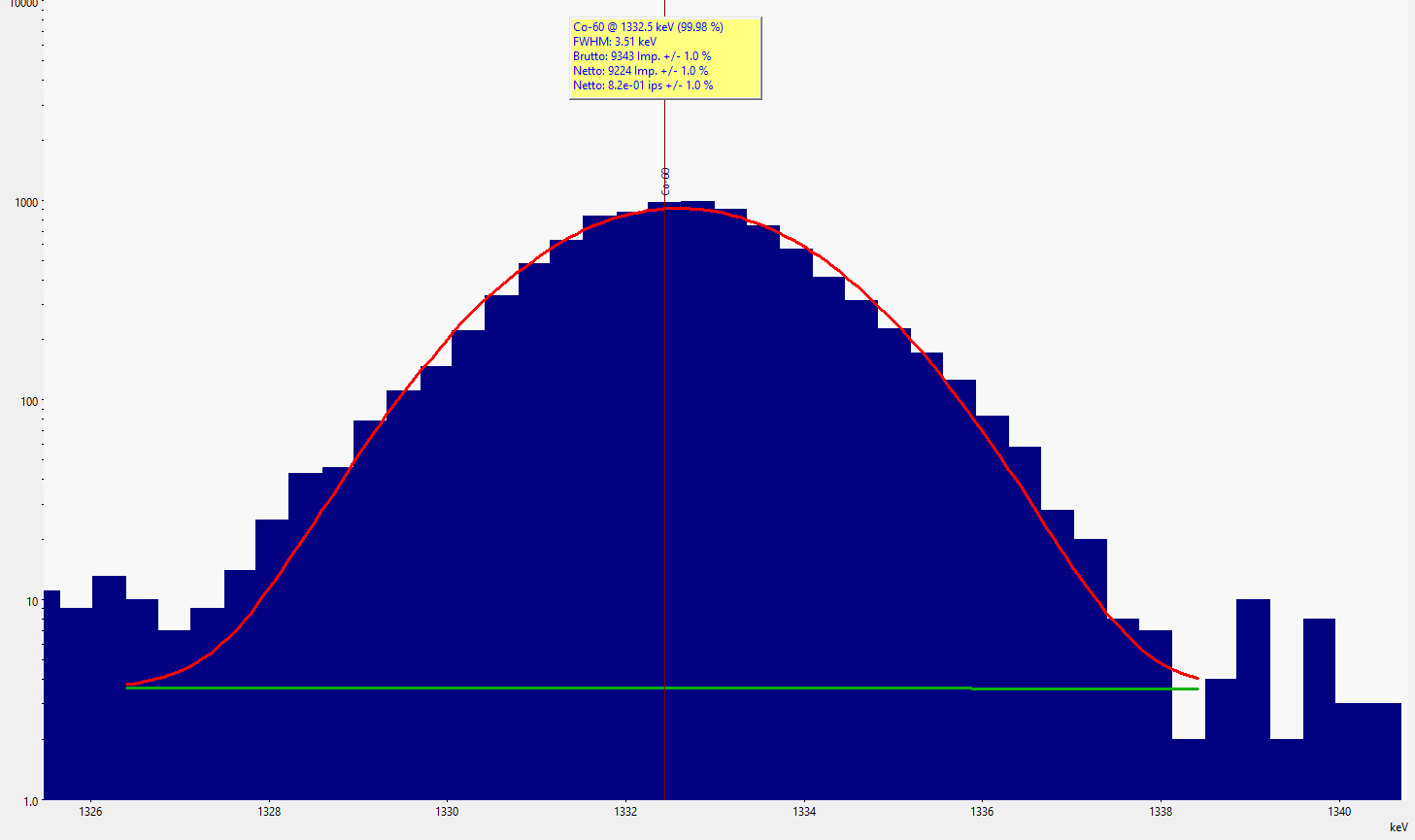
Excerpt of a gamma spectrum with fitted function (red) to the data points of the peak. The green line indicates the assumed background trend. The info box above the peak lists further results of the peak fit, such as the identified nuclide, the energy of the line, the full width at half maximum (FWHM) of the peak, and the total counts (gross) as well as in the peak alone (net) and in the background.
From the shape of the peaks, we can also derive some information necessary for performing correct peak fits:
- Was the peak generated by X-ray or gamma radiation? The so-called X-ray peaks have a different shape (somewhat broader) than gamma peaks.

Comparison of X-ray and gamma peak with similar energy.
- If the peak has a shoulder to the right and/or left of its maximum, this indicates that it is not a single peak but rather two or more peaks that only differ slightly in energy.
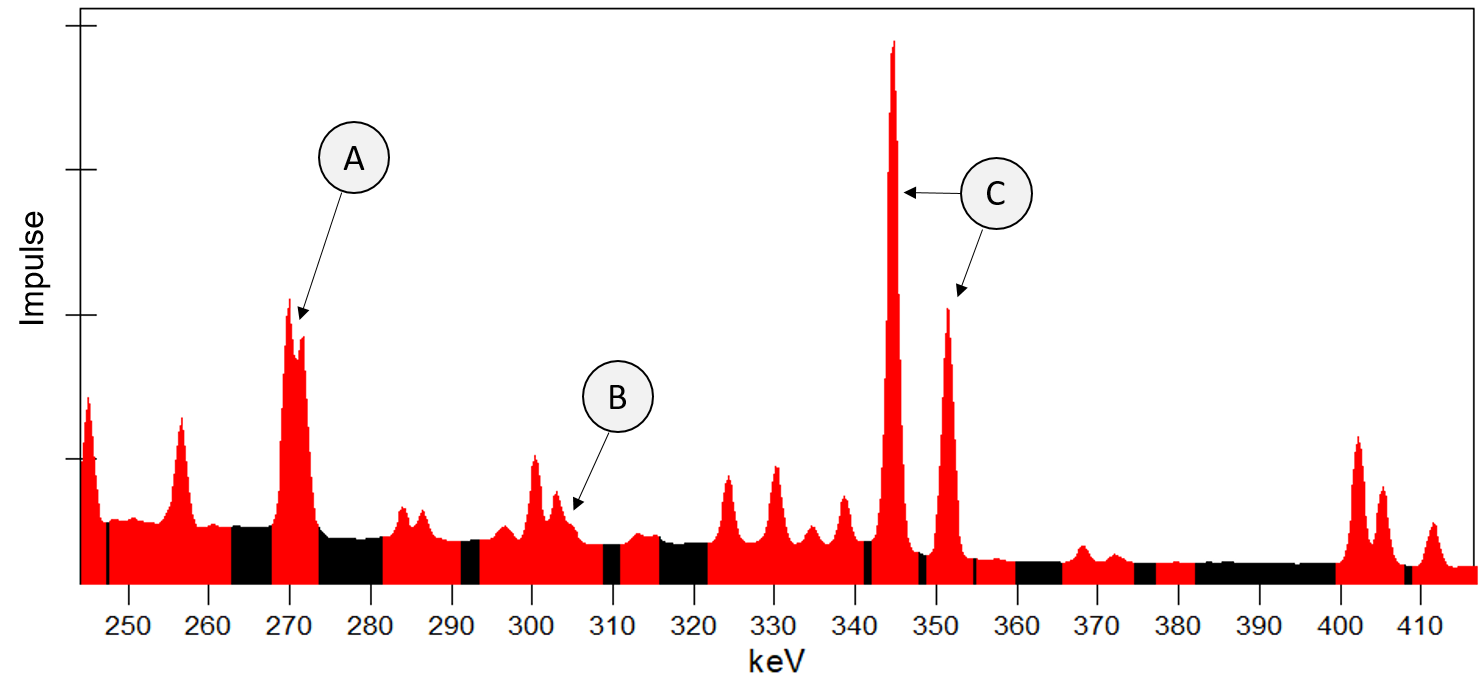
Excerpt of a gamma spectrum with overlapping peaks. The values of the measured spectrum are depicted in black, while the results from a peak search with the Genie 2000 software overlap in red with the measured spectrum. A: Overlap of two peaks; B: Area with several overlapping or closely neighboring peaks; C: two clearly separated peaks.
- Gamma peaks can often be well approximated by a simple Gaussian form. Deviations from this form may occur on the right and/or left side of the peak in the tail region. These are referred to as Low-Energy Tailing and High-Energy Tailing and may need to be considered in the used fit functions to properly determine the peak area.
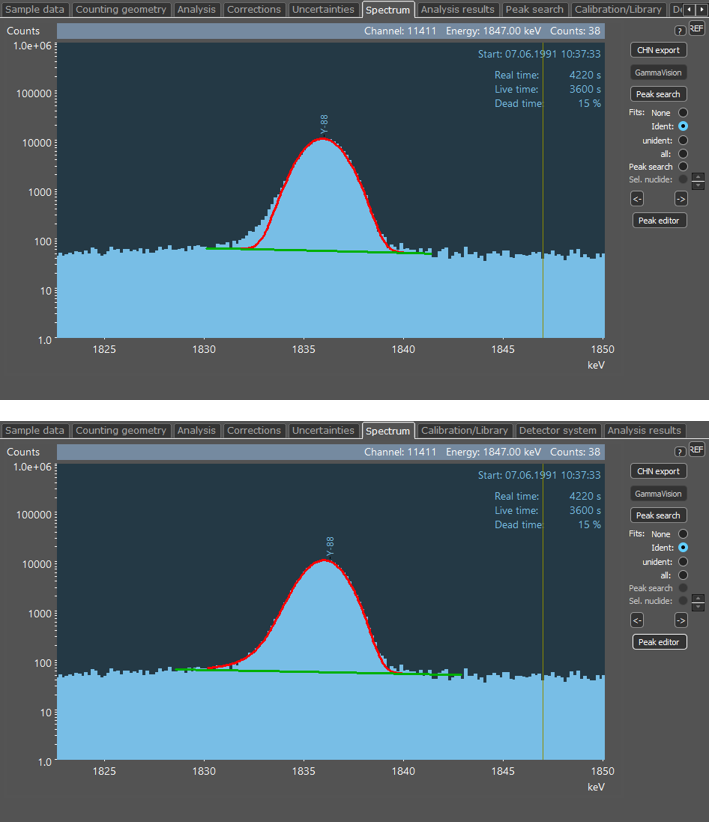
Enlarged representation of a peak (88Y at 1836 keV). Top: Fit of a Gaussian function to the peak (red line). The difference between the measured peak data and the fitted Gaussian function at low energies is clearly visible. This effect is known as Low-Energy Tailing. Below: Fit of an adjusted function to the peak that accounts for both the Gaussian shape of the peak and the Low-Energy Tailing.
Once we have determined the energy values of the maxima (i.e., the highest values of the peaks) for all peaks – including the overlapping ones – we can begin the identification, i.e., assigning the individual peaks to the respective nuclides.
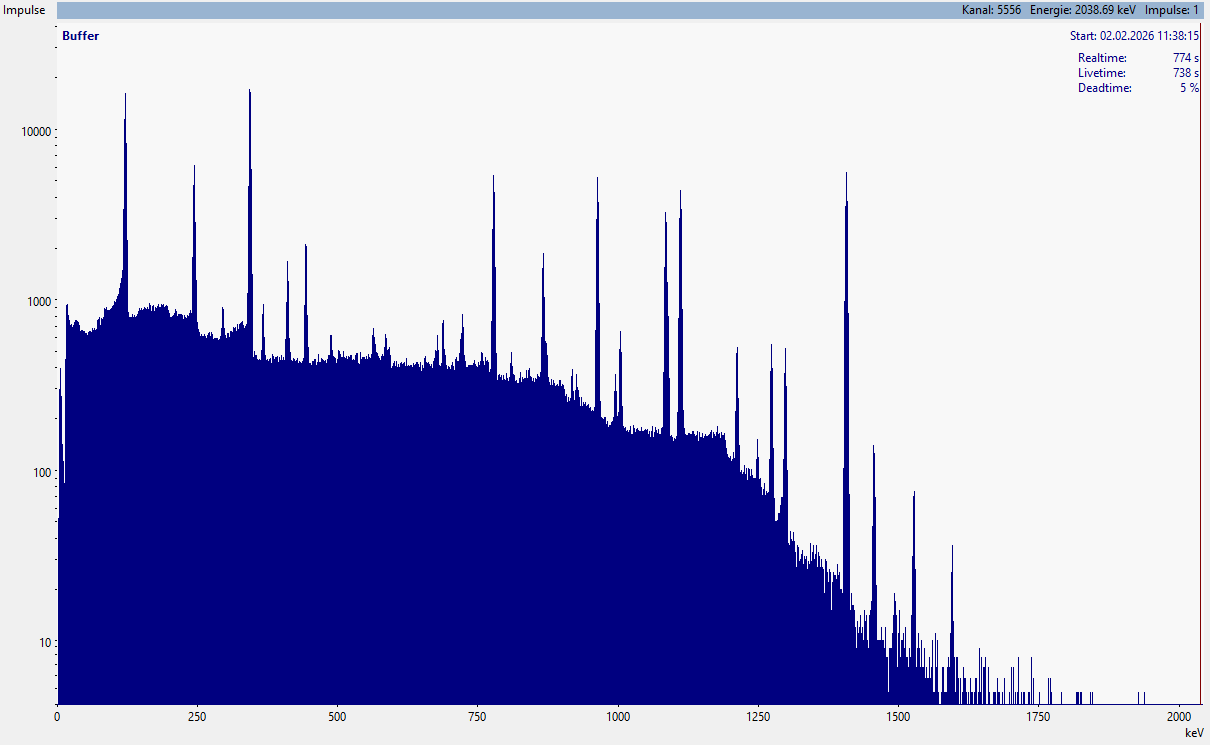
Typical gamma spectrum with numerous gamma lines, which need to be identified all.
For manual evaluation, the following approach has proven effective:
- Start the evaluation with the peak at the highest energy (this is usually the peak that is furthest to the right in the gamma spectrum).
- Use a gamma catalog (e.g., INL) or a corresponding database (e.g., LARA) and search for the corresponding nuclide for this energy.
- Verify that the selected nuclide makes sense. Consider its
- half-life (T1/2) and compare it with the measurement time and any available information on how long the measured sample has existed or, if this information is not available, at least since when it was handed over for measurement. If the half-life is significantly shorter than this time, it is most likely a different nuclide.
- Consider the emission probability of the nuclide at the energy. If this is extremely low, you should consider choosing a different nuclide.
- Check whether the selected nuclide has any additional characteristic gamma lines. Use the gamma catalog or databases for this. Have all of these peaks also been found at the correct energies, assuming their transition probabilities are high enough? If not, you should consider choosing a different nuclide.
- If after the preceding check you conclude that it must be another nuclide, return to point 2, select a nuclide that has a characteristic line at a slightly lower or higher energy (and repeat the subsequent steps. The energy range you should consider, with good energy calibration, is within ± 0.5 eV.
Note:
It is normal for the tabulated values not to match exactly with the measured values. Reasons for this include measurement inaccuracies and statistical errors.
If you believe you have selected the correct nuclide, then repeat the identification at point 2 for the next, still unidentified line with the highest energy.
By the end, you should have identified all peaks of the spectrum. There can be no peak that has not been assigned to a nuclide or an effect!
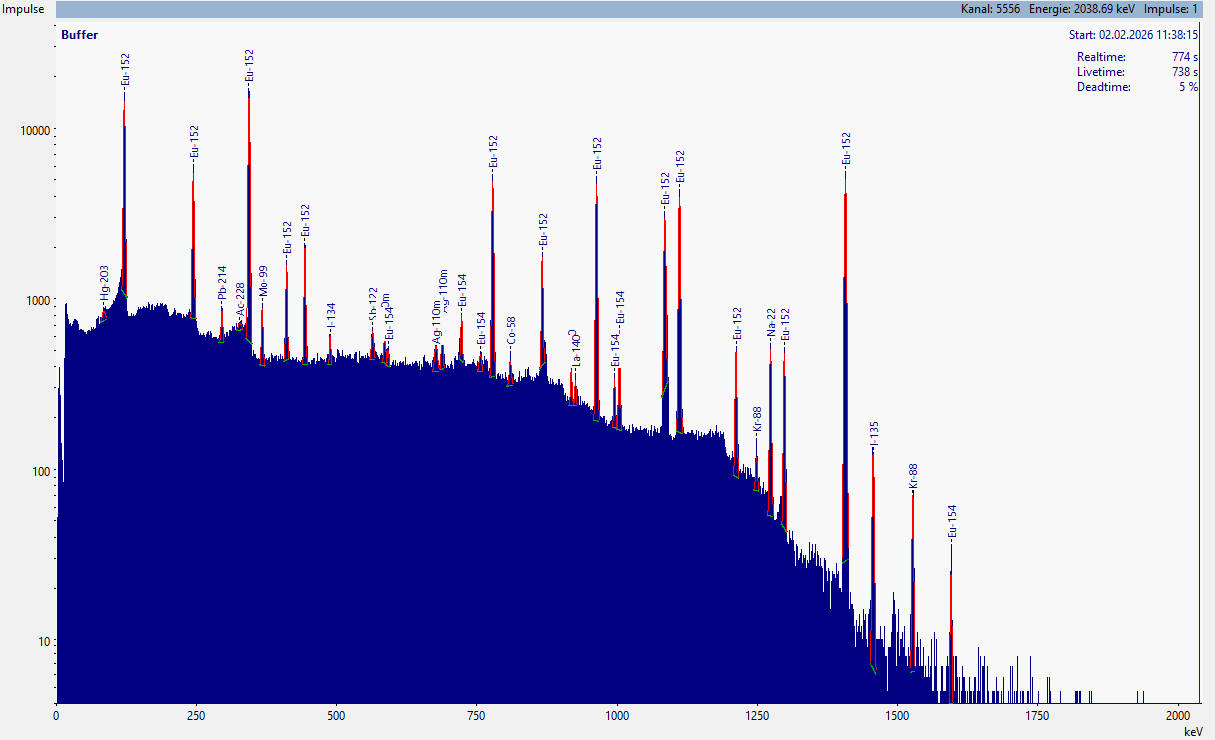
Typical gamma spectrum with all identified gamma lines.
Notes:
The topics of half-life, emission probability, and effects that cause peaks to appear in the spectrum that do not directly correspond to a characteristic line of a nuclide will be discussed in the Advanced section.


